 
Schuster J.C., and Bauer J., The ternary systems Sc-Al-N and Y-Al-N, J. (The arrangement of sulfide ions is identical to the arrangement of chloride ions in sodium chloride.) The radius of a zinc ion is only about 40 of the radius of a sulfide ion, so these small Zn 2+ ions are located in alternating tetrahedral holes, that is, in one. Opposite faces of a unit cell are parallel. This structure contains sulfide ions on the lattice points of an FCC lattice. This is not, however, among the three unit cell criteria: The unit cell is the simplest repeating unit in the crystal. Steckel, Density Functional Theory: A Practical Introduction. In the case of NaCl, the face-centered cubic unit cell has an odd number lattice points and thus doesn't include a whole number of NaCl molecules. These two ions come together to form the lattice. The formula of salt is NaCl, made up of sodium and chlorine ions. 77, 3865 – Published 28 October 1996 Erratum Phys. The regular three-dimensional arrangement of atoms or ions in a crystal is usually described in terms of a space lattice and a unit cell. Perdew, Kieron Burke, and Matthias Ernzerhof, “Generalized Gradient Approximation Made Simple”, Phys. 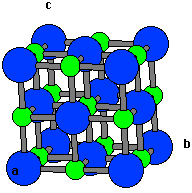
Payne, “First principles methods using CASTEP”, Zeitschrift fuer Kristallographie 220(5-6) pp. Wigner-Zeitz cell consists of all points nearest to a node and has point symmetry of the corresponding Bravais lattice. Only three Bravais lattices with cubic symmetry are shown here. One such factor can be the chosen pseudopotential the use of other pseudopotential types such as LDA can be tested to see if a better comparison between computational and experimental values can be obtained. There are 14 different Bravais lattices in 3D. The difference in lattice constant between the DFT calculated lattice parameter and the experimentally determined value can be attributed to multiple factors. This follows the experimental phase of ScAl, which is found to be the Pm3m space group with a lattice constant of 3.388 Å. From figure 3, it is indeed seen that the CsCl structure for ScAl is favored over the NaCl structure as its ground state energy is lower than that of NaCl. If I remember it correctly, I think NaCl is a cubic lattice where each chlorine atom is surrounded by 6 sodium atoms at equal distance and vice versa. For the BM fit for the NaCl structure of ScAl, the equilibrium lattice constant is found to be 5.663 Å and the bulk modulus is calculated to be 0.111 GPa. Solid lines represent the BM fit and the circles represent the raw DFT data.įrom the BM fit for the CsCl structure of ScAl, the equilibrium lattice constant is calculated to be 3.403 Å and the bulk modulus is found to be 0.450 GPa. Figure 1 shows the CsCl structure.įigure 3: Energy versus lattice parameter for ScAl in the CsCl and NaCl structure. The CsCl structure basis is defined by the space group Pm3m with Cs at (0, 0, 0) and Cl at (0.5, 0.5, 0.5). The aluminum pseudopotential consisted of 3 valence electrons and a cutoff of 2.00 Bohr and the scandium pseudopotential consisted of 11 valence electrons with a cutoff of 1.80 Bohr. The PBE-GGA functional was used in all calculations and the SCF tolerance was set to 2.0e-6 eV/atom. Convergence testing gave total energies converged to 5 meV/atom using a cutoff energy of 600.0 eV and k-point mesh of 10x10x10. The density functional theory (DFT) calculations were conducted using the plane-wave pseudopotential method as implemented in CASTEP. 
In this study, ScAl is tested in both the CsCl and NaCl structures to predict the lattice constant of ScAl in these two forms and it is shown that ScAl prefers the CsCl structure over the rock salt structure. It is a common error to mistake it for a bcc lattice. It has an organized structure and has a 1:1 ratio of Na:Cl. The lattice is cubic primitive with two atoms in the base at (0,0,0) and (,, ). The structure of NaCl is formed by repeating the unit cell. It occurs naturally as white cubic crystals. In the rock salt structure, the two atom types form two interpenetrating face-centered cubic (fcc) lattices. 6.11: Ionic Lattices 6.11B: Structure - Caesium Chloride (CsCl) Rock salt also known as NaCl is an ionic compound. In the CsCl structure, the Cs atoms are in the simple cubic structure and the Cl atoms are in the middle of the Cs atoms cubic structure. \( \newcommand\)).Two common crystal structures of materials with the formula AB are the cesium chloride (CsCl) structure and the rock salt (NaCl) structure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
